North Central London Integrated Care System (NCL ICS) has adopted a proactive, data-driven approach to improving the safety of sodium valproate prescribing for females of childbearing age. Sodium valproate, a medication commonly used to treat epilepsy and bipolar disorder, carries significant risks during pregnancy, including physical and neurodevelopmental harm to unborn children (13 November 2025).
In recognition of these risks, NCL ICS piloted a new set of clinical guidelines alongside a locally developed dashboard to support adherence to national safety priorities. This initiative demonstrates a strong commitment to population health management and clinical safety.
Prior to the release of the Medicines and Healthcare Products Regulatory Agency’s (MHRA) 2023 National Patient Safety Alert, a multidisciplinary working group had already been established. This group included clinicians from medicines optimisation, maternity services, primary and secondary care, and local authorities. An initial audit identified gaps in compliance with MHRA recommendations, such as delays in specialist referrals and inconsistent coding practices in primary care.
In response, the working group developed:
- The North Central London Valproate Risk Minimisation Guideline
- A local dashboard to highlight gaps in care
- EMIS templates to standardise coding and improve clinical workflows
The dashboard was originally built within the HealtheIntent platform, which integrated near real-time data from GP systems and hospital trusts into a unified, longitudinal patient record. Following the decommissioning of HealtheIntent in September 2025, the dashboard was re-designed to make use of the NHS-owned London Secure Data Environment (SDE), offering enhanced security, greater flexibility and improved cost-efficiency.
The valproate dashboard has proven to be a valuable clinical safety tool. It provides a centralised view of the adherence to the Pregnancy Prevention Programme and enables medicines management teams and GP practices to identify and support patients at risk, thereby providing an effective tool to identify gaps in care or coding.

Figure 1. A screenshot of the valproate dashboard displaying NCL’s adherence to national and local safety guidelines that aim to minimise the risk of pregnancy for female patients ≤ 55 years old who are prescribed valproate by their NCL GP.
To better understand the needs of the local population—especially those at risk of health inequalities—additional demographic data was incorporated in the transition to the London Secure Data Environment. This included information such as language, ethnicity, deprivation, and age, enabling identification of specific groups with unmet needs requiring targeted support.
This initiative not only strengthens compliance with MHRA regulations, but also demonstrates how integrated care systems can use data to identify clinical safety incidents in near real-time—ultimately improving patient safety. A built-in notification system automatically alerts the central ICB team when potential safety issues are detected in the data, ensuring GP practice teams are alerted as soon as possible. The dashboard has already enabled timely clinical intervention in a small number of cases, including instances where patients became pregnant while taking valproate.
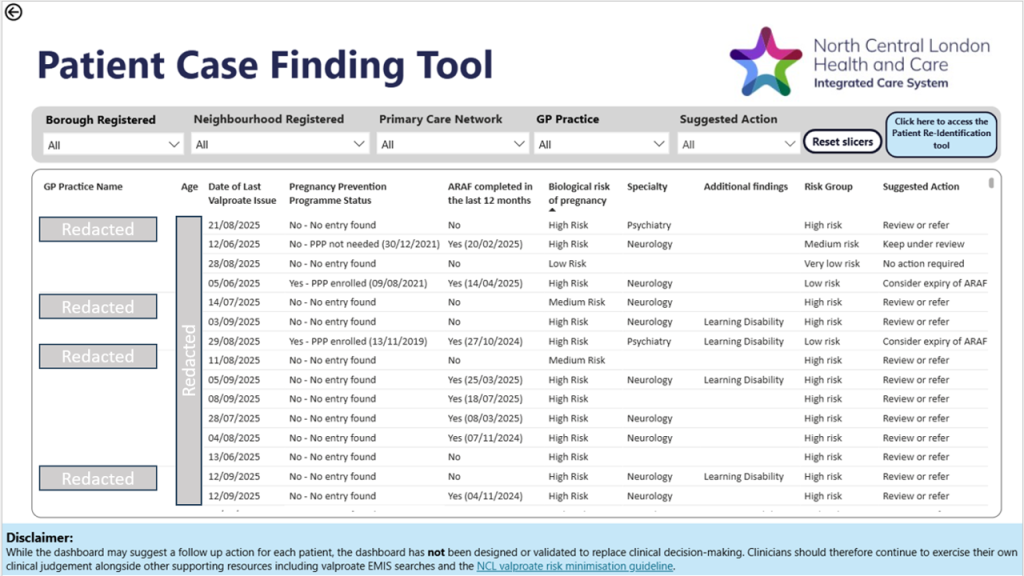
Figure 2. A screenshot of the valproate dashboard displaying the patient case finding tool.
The dashboard is currently in the final testing phase and is expected to go live in November 2025.
You can find out more about the London SDE here.